Molecular mechanisms driving the asymmetric cell division during early embryogenesis of the sea urchin
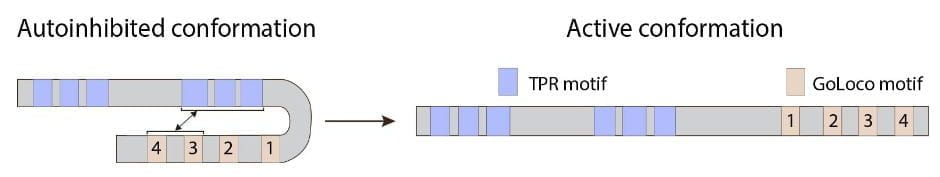
Activator of G-protein signaling (AGS) is a polarity factor involved in the process of asymmetric cell division. AGS is characterized by a two-domain structure with tetratricopeptide repeats (TPR) in the N-terminus and G-protein regulatory motifs (GoLoco) in the C-terminus. While the TPR repeats are involved in protein-protein interactions with AGS binding partners, the GoLoco motifs bind to the heterotrimeric G-protein subunit Gαi to be anchored at the cell cortex. AGS activity is regulated by its conformation: when the TPRs interact with the GoLoco motifs, AGS is autoinhibited until the intramolecular interaction is relieved by Gαi binding.
Asymmetric cell division (ACD) is a critical process for all multicellular organisms as it contributes to cellular diversity and tissue morphogenesis. Through the unequal segregation of proteins such as cell fate determinants, ACD generates daughter cells with distinct molecular profiles and often times unequal sizes.
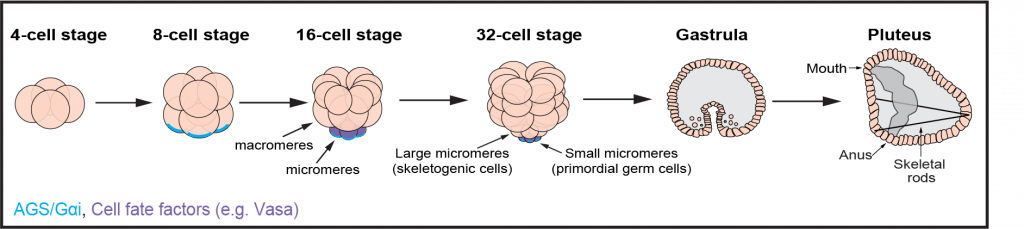
The sea urchin embryos undergoes its first ACD at the 16-cell stage where a new cell lineage is formed called the micromeres. Micromeres have unique features including a smaller size compared to other blastomeres, distinct cell fates and their function as an organizer center that drives endomesodermal specification such as gastrulation.
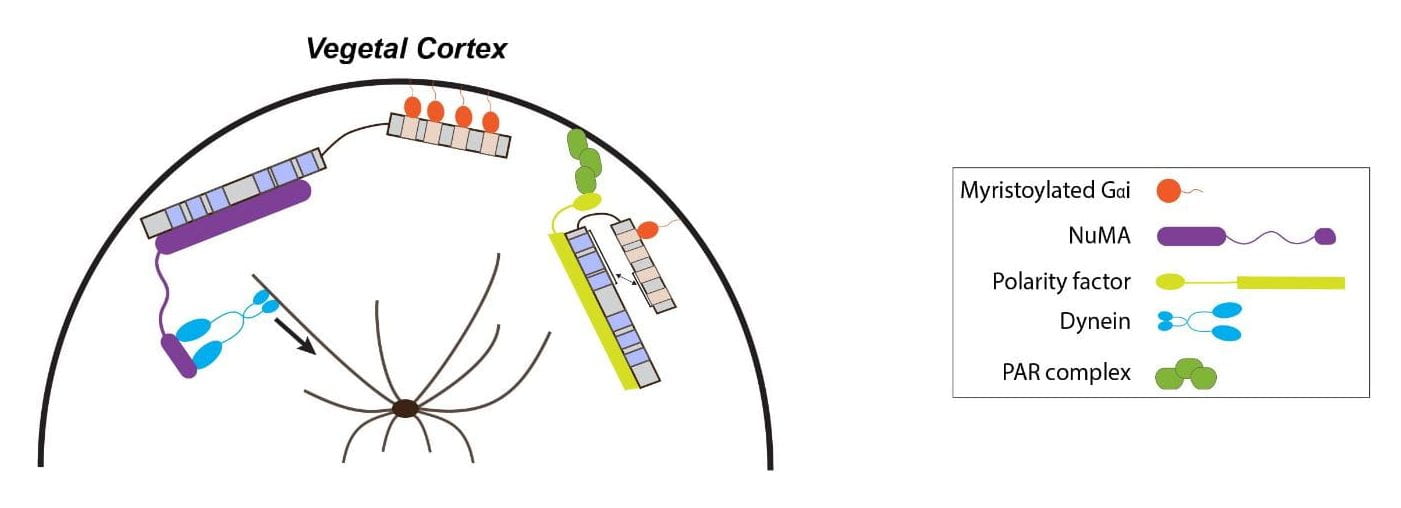
Studies in model organisms such as the nematode C. elegans, the fly Drosophila and mammalian cell cultures have revealed AGS binding partners and mechanisms behind ACD. Briefly, AGS TPR motifs first interact with the polarity factor Inscuteable for recruitment at the cortex where AGS GoLoco motifs bind Gαi, anchoring AGS to the cortex and relieving the autoinhibition. Then, AGS TPR motifs interact with NuMA, which recruits the motor protein dynein to generate pulling forces on the astral microtubules and drive ACD.
In the sea urchin, AGS localizes to the vegetal cortex starting at the 8-cell stage and is critical for ACD and micromere formation. Particularly, the first GoLoco motif is necessary for proper cortical localization of AGS at the vegetal cortex and its function. Further, the TPR motifs help restrict AGS vegetally. We here study further the involvement of each motif found in the sea urchin AGS protein and how modifying these motifs affect AGS localization and function. We also investigate how evolutionary modification of AGS proteins potentially introduced new developmental programs among echinoderms, leading to the formation of a new cell type.
Publications:
- Wavreil FDM, Yajima M. Diversity of activator of G-protein signaling (AGS)-family proteins and their impact on asymmetric cell division across taxa [published online ahead of print, 2020 Jul 18]. Dev Biol. 2020;S0012-1606(20)30198-6. doi:10.1016/j.ydbio.2020.07.004
- Poon J, Fries A, Wessel GM, Yajima M. Evolutionary modification of AGS protein contributes to formation of micromeres in sea urchins. Nat Commun. 2019;10(1):3779. Published 2019 Aug 22. doi:10.1038/s41467-019-11560-8