Glia-Neuron Interactions:
Oligodendrocytes make myelin, a compact, multi-layered, fatty sheath that wraps and insulates specialized neuronal processes known as axons. Myelin’s main function that we know of is that it greatly increases the speed that axons conduct electrical impulses which they use to communicate to one another. However, studies have shown that myelin also has important roles in providing metabolic support to the neurons they wrap, learning and memory, and neuroprotection.
To better understand how oligodendrocytes support neurons, we plan to use mutant mouse strains that have altered myelination, in particular a mouse strain that lacks oligodendroglia and myelin along the optic nerves, which are normally highly myelinated. We plan to examine differences in axon structure and function along unmyelinated axon segments and myelinated segments using super resolution microscopy methods as well as methods that examine local protein production and transport functions along axons.
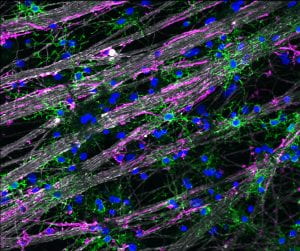
Glia-Glia Interactions:
Oligodendrocyte precursor cells (OPCs) are highly motile and proliferative cells that occupy the entire CNS early in development. At the appropriate time these cells will differentiate into oligodendrocytes that then go on to myelinate axons throughout the CNS, but the cues that signal this differentiation are not completely understood.
Recent research has found that oligodendrocyte differentiation will occur in the developing optic nerve in the absence of dynamic neuronal signaling (Mayoral et al, 2018. Cell Reports), suggesting that the signals that regulate this differentiation are non-neuronal. Therefore we are investigating the role that other cells (ie. astrocytes, microglia, etc.) may play in regulating oligodendrocyte differentiation during development.
We plan to use next generation gene expression analysis methods to uncover the non-neuronal signals that are switched on or off at the time of oligodendrocyte differentiation during development and then use in vitro co-culture systems to screen the signals for roles in promoting or inhibiting oligodendrocyte differentiation. Finally we plan to test the signals in vivo using viral overexpression and CRISPR gene editing technologies.
Selected Publications:
Pan S, Mayoral SR, Choi HS, Chan JR, Kheirbek MA. Preservation of a remote fear memory requires active oligodendrogenesis. Nat Neuro. 2020. 23:487-499. doi: 10.1038/s41593-019-0582-1.
Rankin KA, Mei F, Kim K, Shen YA, Mayoral SR, Desponts C, Lorrain DS, Green AJ, Baranzini SE, Chan JR, Bove R. Selective estrogen receptor modulators enhance CNS remyelination independent of estrogen receptors. J Neurosci. 2019. 39:2184-2194. doi: 10.1523/JNEUROSCI.1530-18.2019.
Mayoral SR, Etxeberria A, Shen, YA, Chan JR. Initiation of CNS myelination in the optic nerve is dependent on axon diameter. Cell Reports. 2018. 16:544-550. doi: 10.1016/j.celrep.2018.09.052.
Mei F, Mayoral SR, Nobuta H, Wang F, Desponts C, Lorrain DS, Xiao L, Green AJ, Rowitch D, Whistler J, Chan JR. Identification of the kappa opioid receptor as a therapeutic target for oligodendrocyte remyelination. J Neurosci. 2016. 36:7925-35. doi: 10.1523/JNEUROSCI.1493-16.2016.
Etxeberria A, Hokanson K, Dao DQ, Mayoral SR, Mei F, Redmond SA, Ullian EM, Chan JR. Dynamic modulation of myelination in response to visual stimuli alters optic nerve conduction velocity. J Neurosci. 2016. 36:6937-48. doi: 10.1523/JNEUROSCI.0908-16.2016.
Mayoral SR, Chan JR. The environment rules: spatiotemporal regulation of oligodendrocyte differentiation. Current Opin Neurobio. 2016. 39:47-52. doi: 10.1016/j.conb.2016.04.002.
Hernandez M, Patzig J, Mayoral SR, Costa K, Chan JR, Casaccia P. Mechanostimulation promotes nuclear and epigenetic changes in oligodendrocytes. J Neurosci. 2016. 36: 806-13. doi: 10.1523/JNEUROSCI.2873-15.2016.
Mei F, Fancy SP, Shen YA, Niu J, Zhao C, Presley B, Miao E, Lee S, Mayoral SR, Redmond SA, Etxeberria A, Xiao L, Franklin RJ, Green A, Hauser SL, Chan JR. Micropillar arrays as a high-throughput screening platform for therapeutics in Multiple Sclerosis. Nature Med. 2014. 20: 954-60. doi: 10.1038/nm.3618.
Full list of publications:
https://www.ncbi.nlm.nih.gov/myncbi/1tSpvj4Eihl/bibliography/public/