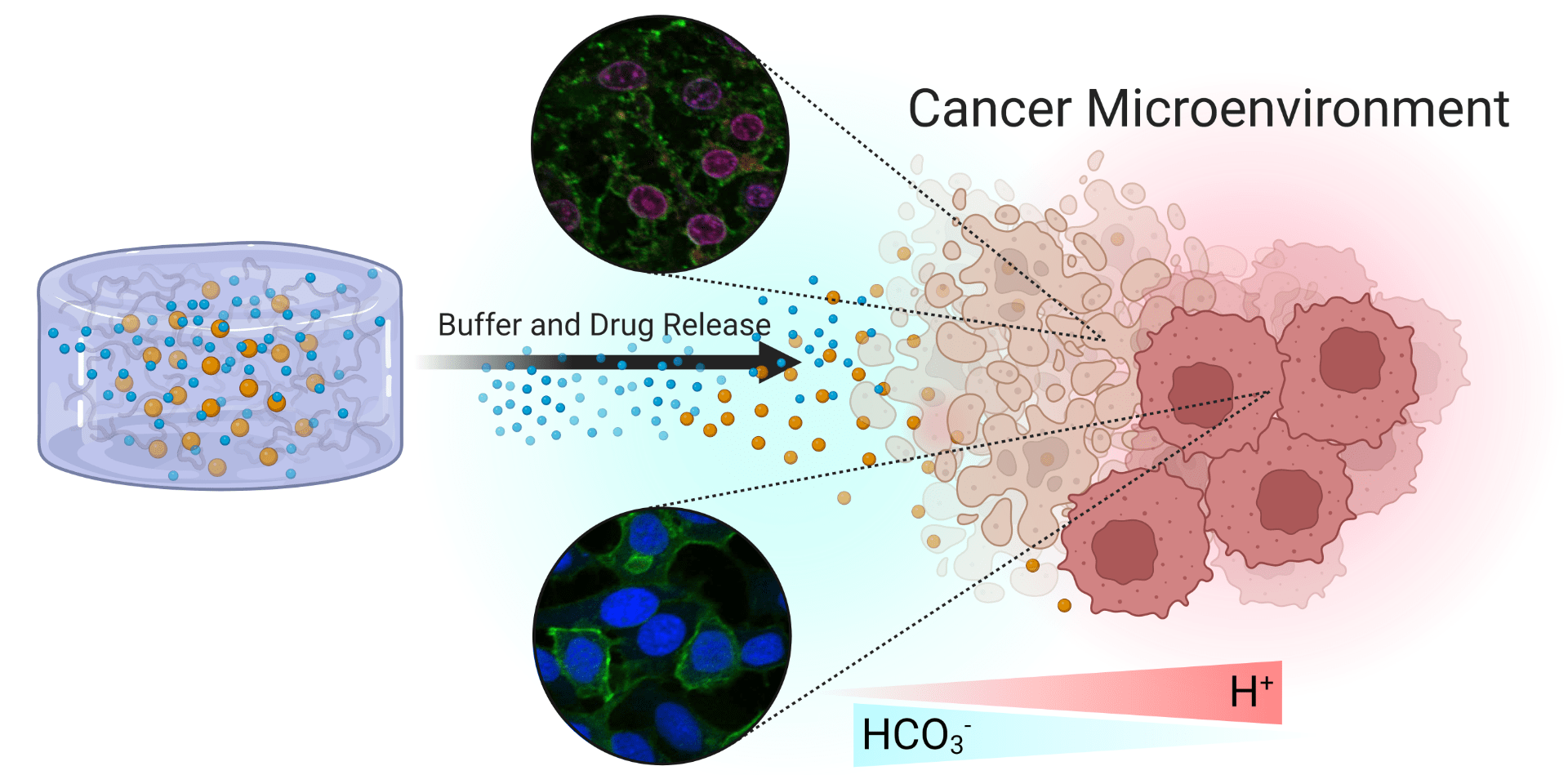
Modulation of pH is crucial to maintain the chemical homeostasis of biological environments. Irregular metabolic pathways exhibited by cancer cells result in the production of acidic byproducts that are excreted and accumulated in the extracellular tumor microenvironment reducing its pH. As a consequence of the lower pH in tumors, cancer cells increase the expression of metastatic phenotypes and chemotherapeutic resistance. We are developing an injectable chitosan-polyethylene glycol hydrogel that can be dual-loaded with doxorubicin and sodium bicarbonate providing alkaline buffering of extracellular acidity and chemotherapeutic delivery to increase chemotherapeutic efficacy. The chemotherapeutic activity of doxorubicin was evaluated in the presence of our pH regulating hydrogels. The findings suggest a promising non-toxic biocompatible hydrogel buffer delivery system that can achieve two simultaneous important goals of local acidosis neutralization and chemotherapeutic release.
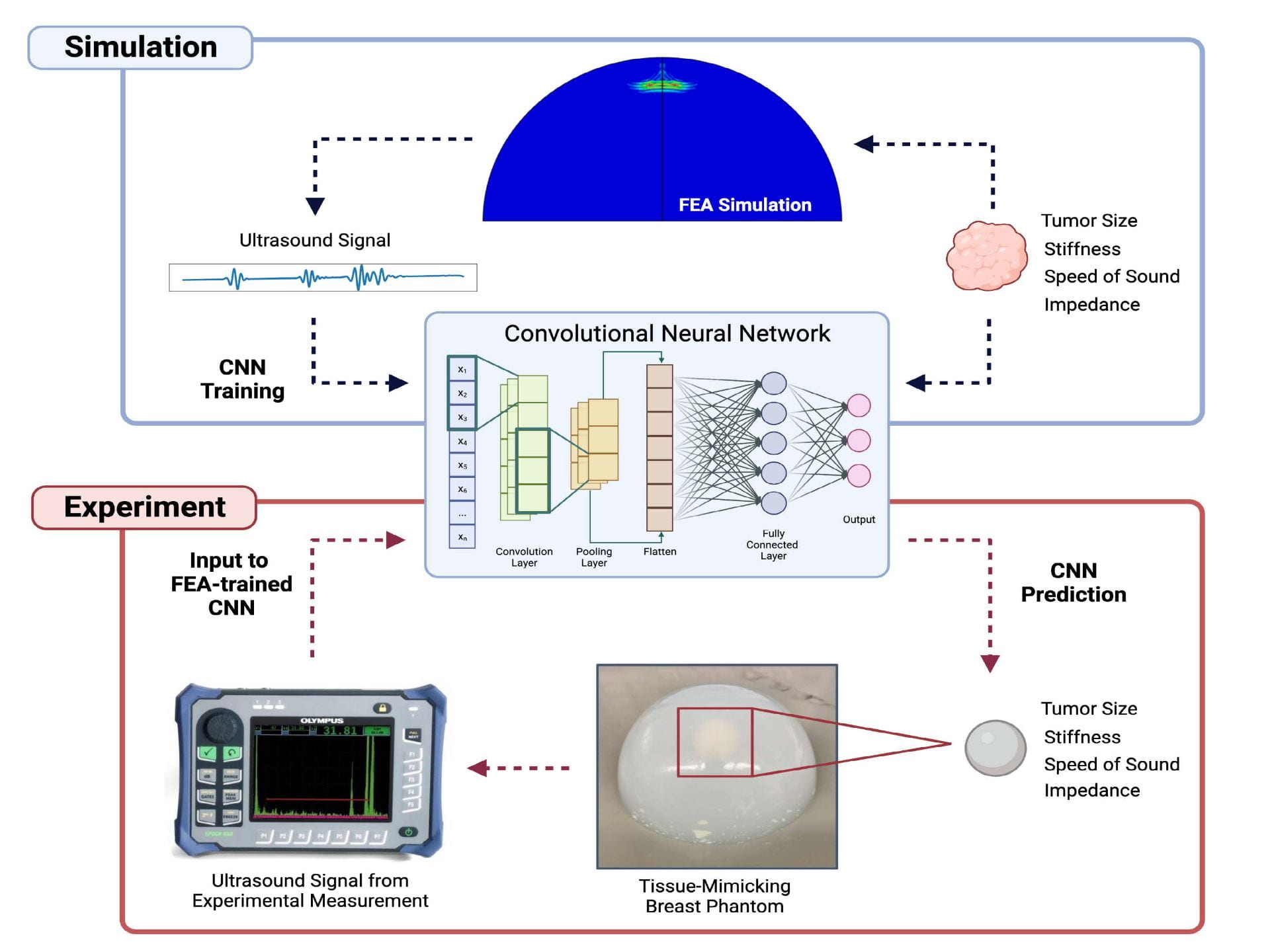
Insights into several unsolved structural integrity problems led us to develop an evolving research area that integrate advancements in neural networks with mechanics to solve critical structural integrity problems. Crack-driven fracture and plasticity-based collapse are two key structural failure mechanisms. Our research on neural networks with mechanics is divided into the following two areas (i) computational simulations data trained convolutional neural networks (CNN) for accurate hidden crack quantification (and interacting flaws) and (ii) physics-informed neural networks (PINN) for boundary value finite strain plasticity problems. The first approach relies on data to train a CNN, while the second approach with PINN does not require data for training. We have recent publications on CNN to characterize hidden flaws and PINN for large strain plasticity problems. We are developing neural network methods for early detection and quantification of breast cancer tumors using ultrasound.

Dynamically crosslinked polymers are strongly rate stiffening materials that are gaining significant interest due to their potential energy absorption and self-healing abilities, and have the promise to be used for sensing, actuation, and lightweight body or structural protection. These soft polymers and gels change from viscous-fluid-like to rubbery and glassy-solid states during high-rate deformations due to the microstructural mechanics of reversible, dynamic crosslinks. We are studying the rate-dependent deformation and fracture of this class of soft polymers and their composites and developing continuum scale models.

Fiber-reinforced polymer (FRP) composites have become integral to a wide variety of structural applications, ranging from aerospace components that require optimal weight-to-strength ratios to civil infrastructure projects such as bridges and buildings, where corrosion resistance and durability are of the utmost importance. However, prolonged exposure to extreme environments can compromise the structural integrity of the composite. In this collaborative project, we are developing a multiscale damage model that encompasses a range of degradation mechanisms occurring at different scales, like fiber-matrix debonding, micro-cracking, delamination, and chemical interactions, arising due to the hygrothermal effects.
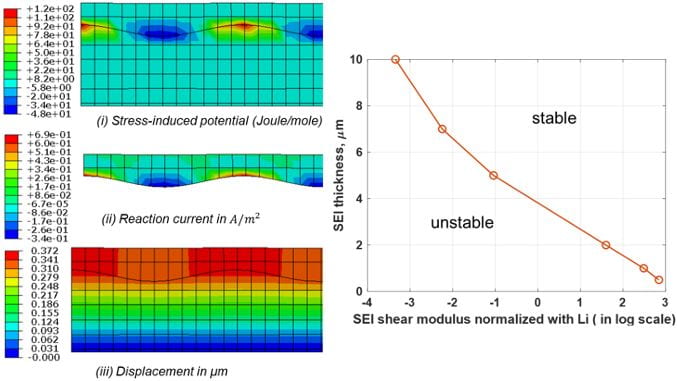
Lithium (Li) metal is an ideal Li battery anode due to its high theoretical specific capacity and the lowest negative electrochemical potential. However, Li-anode batteries have seen limited success due to dendrite growth and solid electrolyte interphase (SEI) failure. We are studying coupling between stress and electrochemistry and developing integrated electro-chemo-mechanical models and computational methods to predict Li-anode and SEI instabilities and failure mechanisms
Recent past
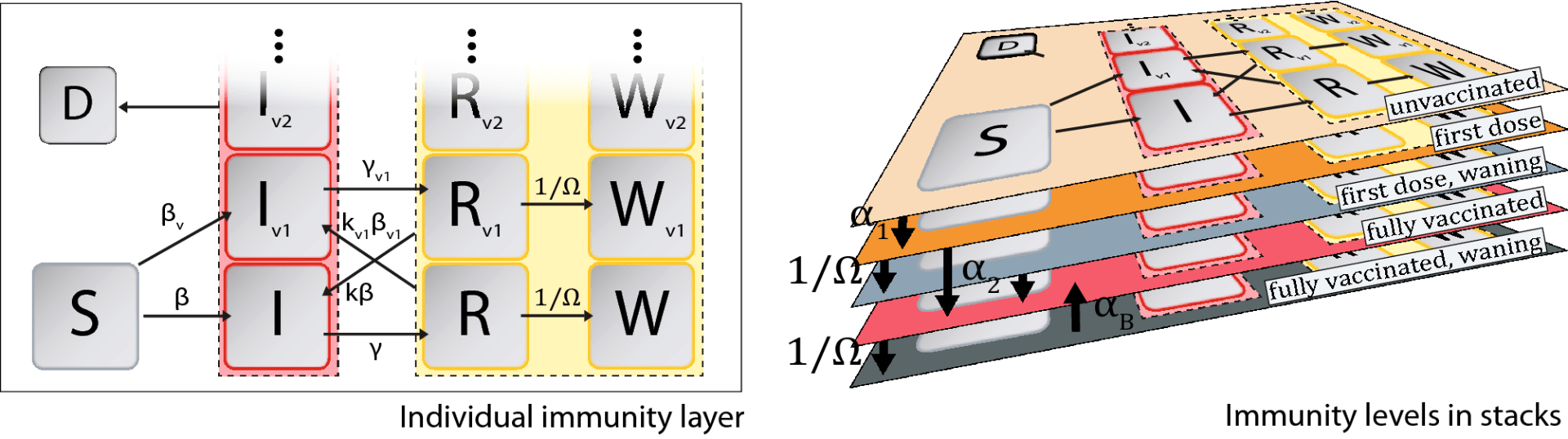
We developed a SIR model that includes population behavior – level of caution due to increasing infection and a sense of safety due to vaccine availability to explain and capture the multiple infection case peaks observed during the multi-year course of COVID-19. We also developed a model to predict coupled effects of vaccine multiple-dose roll-out strategies, vaccine efficacy, waning immunity, population level of caution, sense of safety, under-reporting of cases, and highly prevalent variants.
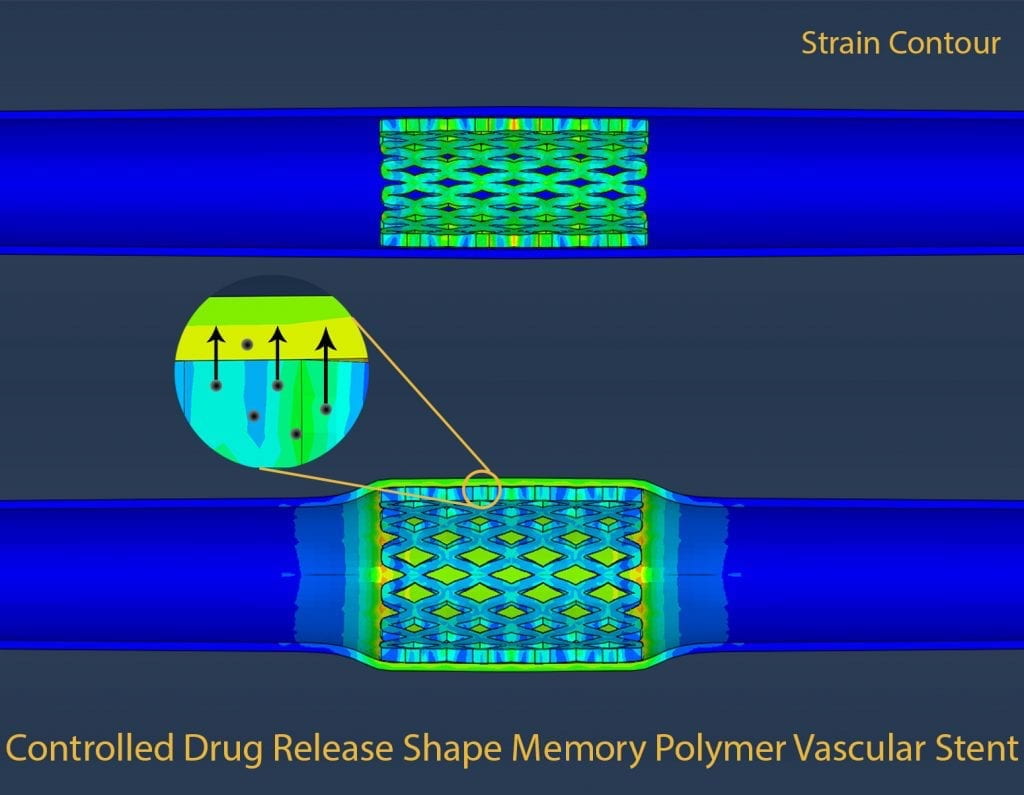
Shape memory polymers and metal alloys are responsive materials that are able to recover large strains and their original shapes from temporary shapes. The use of biocompatible shape memory polymers and alloys is useful in minimally invasive surgeries where there are limitations to access due to anatomical considerations. We are developing shape memory polymer and alloy based novel minimally invasive biomedical implants.
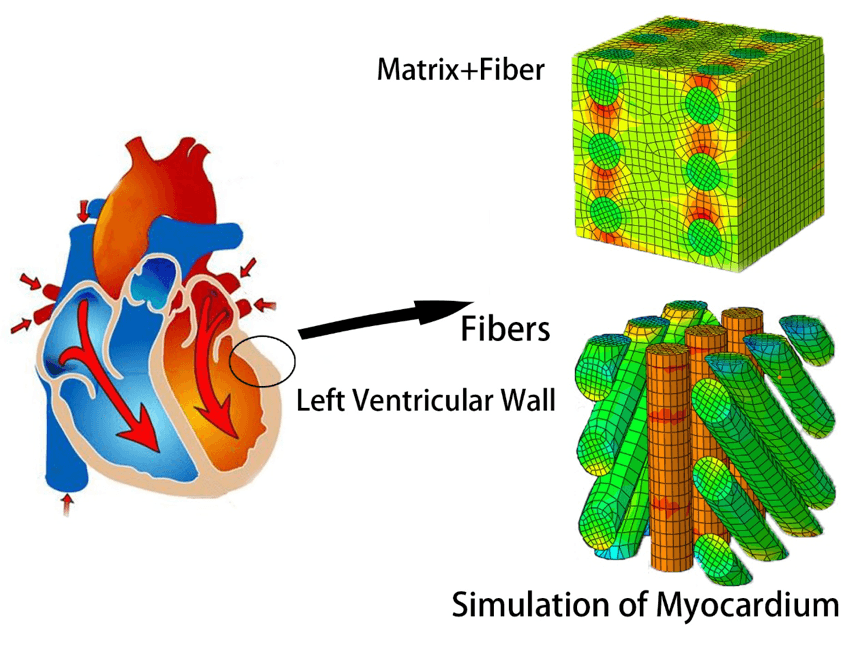
Cardiac patch therapies have promise to restore the heart function and lower the risk of heart failure after myocardial infarction. We have developed a continuum scale anisotropic model and finite element computational tools for the anisotropic response of engineered composite microarchitecture tissue scaffolds. Our model could allow design and analysis of optimal engineered scaffold tissues.
